by Gordon S. Johnston, Professor of Biology, Nasson College, August, 1973
Updated November, 2019
Introduction
I first saw the pond in the company of students John Holzapfel and Sidney Korn on a cold January morning in 1966. It was an impressive introduction for the ice was groaning. Strain cracks were streaking across the ice from one side of the pond to the other, accompanied in their passage by groaning and renting sounds. During the years 1967 to 1978, with the help of innumerable students, I have witnessed the progress of the seasons at the pond, probed its shores, plumbed its depths, and reflected on its past. Of all of the things that we have learned about the pond, the one aspect that is most meaningful to me is that I now comprehend Henry Thoreau’s love and fascination for Walden Pond. To me, Deering Pond is another Walden.
History
That body of water presently known as Deering Pond came into existence sometime before 13,350 years ago during the retreat of the Wisconsin Glacier1. The basin in which the pond lies was gouged out of the metamorphic rocks of the Rindgemere formation by earlier glacial advance2. As the ice mass melted, a depression ringed with glacial debris and paved with rock flour was left behind to catch water flowing from the hills and ridges in the vicinity. The original depth of the basin has not been ascertained, but superficial investigations suggest that it must have been at least seventy-five feet deep with a water level several feet higher than at present. Such was the birth of Deering Pond.
Within a short period of time, the inevitable aging process which afflicts all standing bodies of water commenced. The shallowing process was heralded by the arrival of Sphagnum moss and the growth of plants in the marginal water of the western shore. The growth of these plants was encouraged by the acidic minerals in the surrounding till and bedrock, while at the same time, decay of the organic material was retarded. Consequently, peat, the product of partially decayed organic material, gradually began to accumulate and fill the basin. Analysis of a peat core made by an ecology class in the fall of 1972 indicates that acid-loving diatoms were present quite early in the history of the pond and have predominated ever since. This substantiates suspicions that Deering Pond was predisposed to rapid filling.
The first human to view the pond undoubtedly was an Indian for early maps of the area give the name of the pond as Tombegewoc3. Huden4 states that this is an Abenaki word meaning “place of the rocky reef in the pond”. The presence of boulders at or below the water surface support this description. On the other hand, Eckstom5 contends that the word signifies “rapids” and refers to a rapid on the outlet. Since the stream leaving the pond flows smoothly for some distance, this translation does not seem to apply. Whatever the case may be, the Indians apparently were familiar with the pond and utilized its fish and plants.
There is no available record concerning the first colonist to view the pond. Abraham Preble passed within a half mile of it in 1720 when he was establishing the boundaries of the township3. However, he probably did not see the pond owing to the tree growth which existed at the time. John Deering inherited the land surrounding the pond from his grandfather who previously was granted the land by the Crown for his services in the French-Indian War of 1693. William and Gideon Deering, sons of John, settled about one mile northwest of the pond around 1789. Hence, the pond, Deering Ridge, and the Deering Neighborhood are named for his family. Under a colonial ordinance made at the time Maine was part of the Colony and Province of Massachu- setts Bay, the State of Maine today has jurisdiction over the pond itself.
The presence of stonewalls and barbwire fences indicates that most of the land around the pond was eventually cleared. Rev. Ilsley6 reports in 1872 that the largest mast pines in the State were cut on a lot west of Deering Pond and that the stumps were still present at that time.
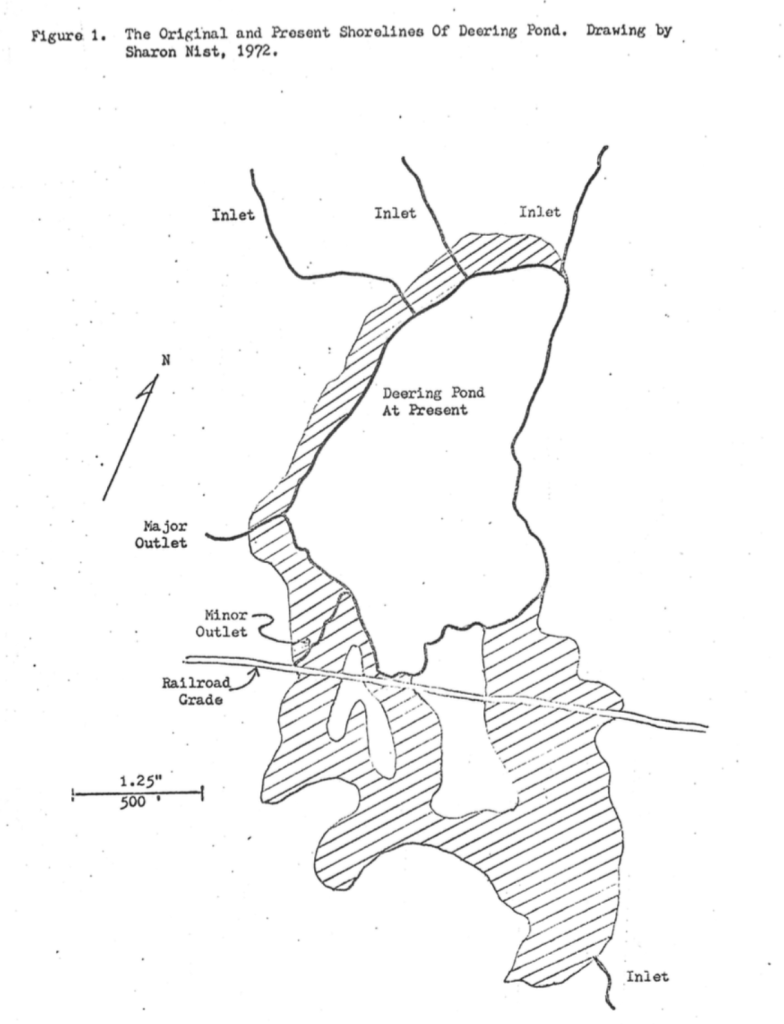
Human activities up until the years of 1870-1871 apparently had but little influence upon the pond. However, the construction of the Portland and Rochester Railroad (later the Sanford and Eastern) brought about significant alterations in two original sources of water to the pond3.
On the southwest side of the pond, the railroad grade was built across three boggy areas which formally were part of the pond in 1871. Drainage from a marsh to the present pond was blocked, but not without retribution. At the first of these boggy areas, a flat car, standing by itself sank out of sight overnight at the foot of the pond in May of 18713.
On the southwest side of the pond, the grade intercepted the third area that had an inlet stream. The stream flowing from Hanson’s Ridge was diverted into a ditch on the south side of the grade while the former mouth became an outlet of the pond flowing down the north side of the same grade. These modifications of the drainage patterns almost a century ago probably lowered the water level of the pond and stabilized it against seasonal oscillations. In turn, the new water level favored more rapid filling of the basin with organic material.
Mr. A. J. Rollins of Nasson College secured interesting documentation of the railroad construction in the form of an increment boring taken in 1971 from a large red maple that was growing adjacent to the railroad grade. As a seedling, it had established itself when the land was cleared. For several years it grew vigorously until it was crowded and shaded by other trees that also had invaded the site. It grew slowly for approximately a half a century until 1938, at which time vigorous growth was renewed. Apparently, the hurricane of 1938 blew down the surrounding trees allowing the red maple to stretch in the sunshine.
Within the last sixty years, logging operations and a fire in 1953 have occurred on both the eastern and western sides of the pond. The creation of logging roads may have lowered the water level of the pond somewhat, but this was offset by beavers building a dam on the major outlet in the early sixties. These beavers vanished around 1967 and the pond level fell again. In 1971, the beavers re-colonized the pond and established the present day water level. Both the fire and logging have left their marks in the form of mosaic patterns of vegetation that are in various stages of succession.
Present Day Conditions
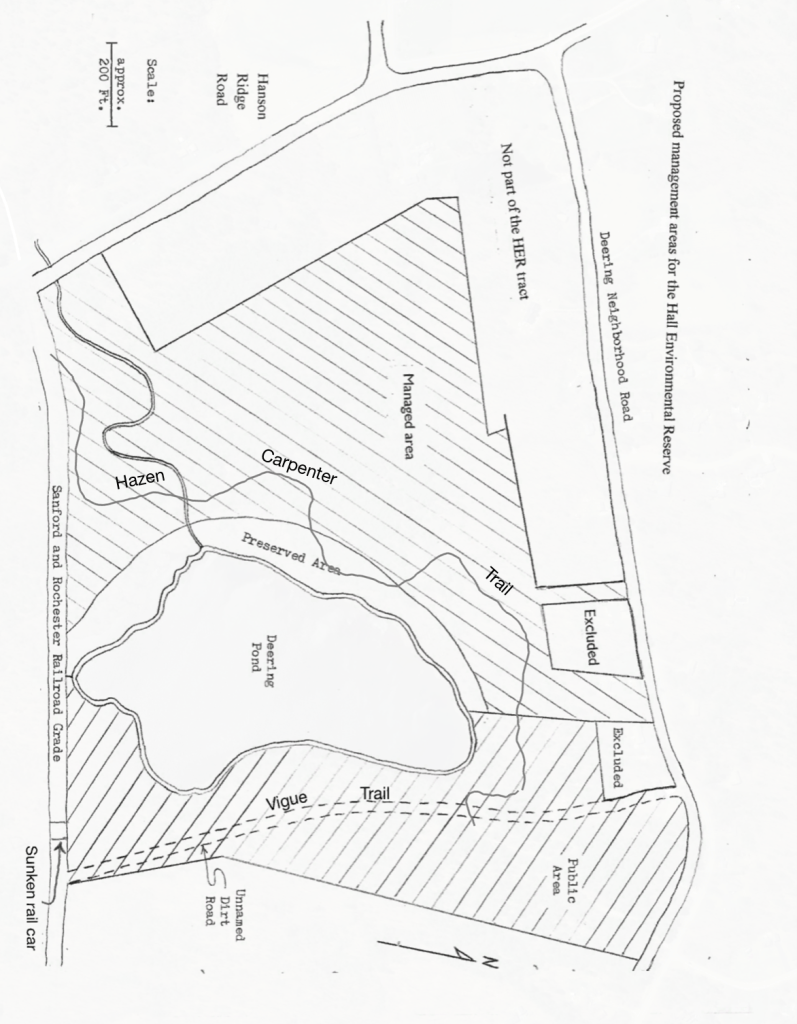
The land around the pond was owned by Nasson College from 1972 to 1983. It was designated as the Russell Environmental Study Tract in honor of Mrs. Ina Russell who purchased the land from Veterinarian Dr. Ralph Vigue and donated it to the College. The area served as a wildlife preserve, an outdoor classroom, and as a “green space”. In 1999 the Hall family of Cambridge, Massachusetts, who purchased it at auction, donated this property to the Sanford Springvale Mousam Way Land Trust. Today The Hall Environment Reserve at Deering Pond is a signature holding of the Sanford Springvale Mousam Way Land Trust with the Sanford Railroad Trail on the south, the much improved Vigue Trail on the east, and the Hazen Carpenter Trail circling the west and the north.
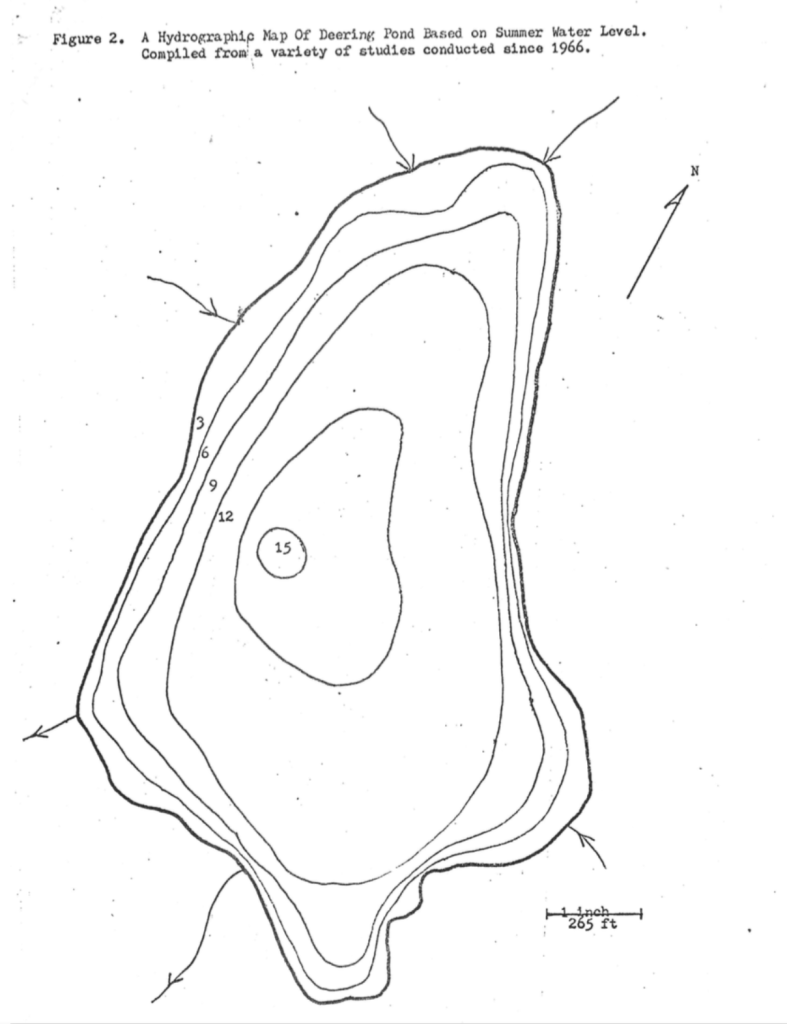
Deering Pond has a surface area of twenty-six acres, a shoreline of approximately 0.8 of a mile, and an average depth of 7.5 feet. The bottom, which is composed of unconsolidated colloidal ooze, is fifteen-feet below the surface at its deepest point. This ooze has the remarkable property of forming a gel upon prolonged exposure to the air. In the course of a field trip some years ago, a bucket of sediment was brought back to campus. Somehow the bucket was overlooked for the better part of a year. When it was finally discovered, the contents had acquired the characteristics of “Jello”. Even though the bucket was turned upside down, the gel remained in place.
Microscopic examination of the pond sediment has revealed an abundance of diatoms, pollen grains, and sponge spicules in a matrix of granular colloidal floc. Less common components of the sediment include pieces of plant debris and the exoskeletons of insects. On the basis of preliminary study of the microbiology of the sediment, it appears that floc-forming bacteria of the genera Bacillus, Zooglea, Flavobacterium, and Achromobacter, among others are responsible for the production of vast quantities of colloidal floc present in the basin7. In addition, Sarcina and a species of green sulfur bacterium have been tentatively identified in sediment samples.
Chemical analysis of the sediment has revealed that more than 56 tons of calcium, 1,300 pounds of potassium and magnesium, and 250 pounds of phosphorus are deposited within the sediment. Moderate amounts of nitrogen particularly in the ammonia form have been detected. The pH of the material ranges from 4-5 and, thus far, has proved to be uniform throughout the sediment mass.
The characteristics of the sediment outlined above indicate that this material exerts a prevailing influence upon all living organisms found within the basin. Suspended colloidal material and liberated tannic and humic acids impart a tea color to the water and reduce the depth of light penetration. The average secchi disc depth is around 7 feet.
Furthermore, these materials maintain the acidity of the water which retards, but does not inhibit, the decay activity of micro-organisms. Consequently, near the bottom of the sediment, dissolved oxygen is virtually exhausted while the carbon dioxide level is quite high, a situation that favors the activity of anaerobic organisms such as the reducing and sulfur bacteria which release methane and hydrogen sulfide respectively.
Because nutrients such as nitrogen, potassium, phosphorous, calcium, magnesium, etc. are either bound within the undecomposed matter on the bottom or precipitated out of solution in a colloidal complex, the concentration of dissolved substances in the water is extremely low. Thus, the following limiting factors are imposed upon organisms:
1. A low pH; 2. Low nutrient levels; 3. Low dissolved oxygen levels; 4. Warm water temperatures (summer).
Flora and Fauna
The limiting factors listed above constitute what is known as a dystrophic condition8, 9. Since these factors limit the variety of organisms which can exist, the flora and fauna of bog ponds are impoverished. A partial biotic inventory of Deering Pond is listed below.
Aquatic Plants and Animals
Phytoplankton – quite abundant and relatively diverse: Blue-green algae; Green algae – particularly desmids; Euglenoids; Dinoflagellates; Golden algae – particularly diatoms, that are most abundant.
Zooplankton: Protozoans – particularly sarcodinids; Rotifers – most abundant; Water fleas – particularly Daphnia & Bosmina.
Copepods – particularly Cyclops
Periphyton – attached forms: Blue-green algae – particularly Nostoc; Green algae; Red algae – particularly Batrachospermum; Sponges; Rotifers; Bryozoans.
Worms – flatworms, oligochaetes, and leeches common
Molluscs – quite rare
Insects – mature and immature forms:
Orders: Odonata – dragon and damsel flies; Hemiptera – water bugs; Trichoptera – caddis flies; Diptera – flies and mosquitoes.
Reptiles – mostly regional turtles – particularly painted and snapping.
Amphibians – frogs quite abundant, a few salamanders.
Fish – warm-water species: Suckers, Shiners, Yellow Perch, Minnows, Sunfish, Bullheads, Pickerel, Small-mouth bass introduced in the 1980s.
Floating Aquatic Plants – water lilies, water shield, floating heart, and bladderwort.
Emergent Aquatic Plants – spike rushes and cattails
Bog Plants and Animals
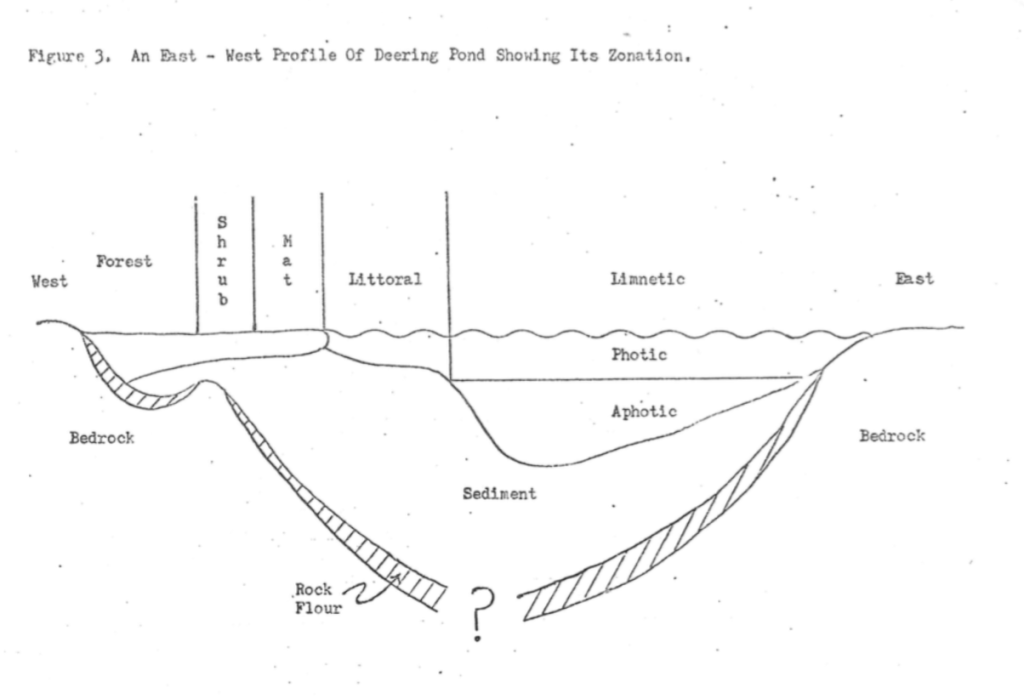
Plants and animals of the bog proper are quite interesting because they represent a subarctic assemblage that is tundra-like9. Bog soils and sphagnum moss are poor conductors of heat. Consequently, such soils or areas covered with sphagnum moss are generally colder than surrounding soils or areas. The following zones or plant communities are present (cf. Figure 3):
Sphagnum mat and heath – at margin of water: Insectivorous plants, Sphagnum moss, Leather leaf, Bog rosemary, Sweet gale, Cranberry (large and small-fruited species), Cotton grass, Orchids, Bog aster, Buttonbush, Purple pitcher-plant.
Shrub: Sheep laurel, Rhodora, Winterberry, Speckled alder, Mountain holly, Bebb’s Willow, Black spruce, Highbush blueberry, Black chokeberry
Woodland: Red spruce, Red maple, Hemlock, Yellow birch, Black gum (rare), Red Spruce.
Animals are not very abundant in a bog and many animals that are found in this habitat do not live there. Some of the animals that have been observed or captured in the bog are:
Spiders and ticks
Insects: Midges, Blow flies, Crane flies, Pitcher plant mosquito, Bumblebees.
Birds: Oven bird, Water thrush, Great blue heron, Marsh hawk, Coot, Tree swallow.
Rodents: Snowshoe hare, Meadow vole, Deer mice, Bog lemming, Water shrew, Red-backed vole
The Future
Eventually Deering Pond will pass from existence and be replaced by a wet woodland. The rate at which this transformation occurs will depend upon the activities of man and beavers. Additions of sewage or septic tank effluent will provide nutrients which are presently lacking. Algal blooms and rapid accumulation of organic material will result from this enrichment. The continued presence of beavers will ensure higher water levels which retard the rate of shallowing and bog mat encroachment. At the same time, their activities will protect the bog from fire damage.
Well-meaning people have, at various times, suggested that the filling process could be delayed by either chemical treatment or dredging. On the surface such actions to preserve the pond seem reasonable. However, chemical treatment or dredging undoubtedly would have serious side effects. With respect to chemical treatment, clarification of the water would encourage extensive growth of the littoral vegetation which is presently limited in extent by low light levels at its outer edge, while destruction of the sediment floc would release large quantities of dissolved substances that could create downstream problems as well as encourage the growth of littoral vegetation.
On the other hand dredging would result in the deposition of a considerable quantity of colloidal ooze in some inappropriate place. In view of the unique properties of this material, disposal of it would be a technological challenge.
Epilogue
The fact that the pond is destined to vanish three or four thousand years from now is saddening. However, it is the natural scheme of things on the grand scale of time.
LITERATURE CITED
1. Neil, Craig D., 1997. Surficial Geology of the Sanford 7.5Minute Quadrangle, York County, ME. Maine Geological Survey Open-File Report 97-70. A radiocarbon age on the bottommost organic sediment was 13,350 +/- 75 years.
2. Hussey, A. H., 1962. The Geology of Southern York County, Maine. Special Geological Study Series, No. 4, Dept. of Economic Development, Augusta, Maine.
3. Prosser, A. L., 1968. Sanford, Maine: A Bicentennial History. The Sanford Historical Society, Sanford, Maine. Note: David Parent, Superintendent of the Sanford Water District, and Gordon Johnston located the actual position of the car with a magnetic pin detector. Journal Tribune, October 4, 2018.
4. Huden, J. C., 1962. Indian Place Names of New England. Museum of American Indians, Heye Foundations.
5. Eckstorm, F. H., 1941. Indian Place-names of the Penobscot Valley and the Maine Coast. The Maine Bulletin, XLIV (4).
6. Ilsley, G. B., 1872. Sanford. In Atlas of York County, Maine: 367. Everts and Peck, Philadelphia, 1880.
7. Dohrman, M. E., 1972. Isolation and Identification of Microorganisms from Deering Pond. Reading and Research Manuscript.
8. Daubenmire, R., 1968. Plant Communities: A Textbook of Plant Synecology. Harper and Row, Inc., New York.
9. Deevey, Edward S., Jr., 1951. Life in the Depths of a Pond. Scientific American, 185:68-72.


4 responses to “The Natural History of Deering Pond”
[…] The Natural History of Deering Pond, by Dr. Gordon S. Johnston, Professor of Biology, Nasson College, August, […]
LikeLike
[…] The Natural History of Deering Pond, by Dr. Gordon S. Johnston, Professor of Biology, Nasson College, August, […]
LikeLike
[…] Pond – By Kevin McKeon, Director, Mousam Way Land Trust, Adapted from The Natural History of Deering Pond 1 By Gordon “Bud” Johnston. And the footnote should have been listed as follows: 1. The […]
LikeLike
[…] Pond – By Kevin McKeon, Director, Mousam Way Land Trust, Adapted from The Natural History of Deering Pond 1 By Gordon “Bud” Johnston. And the footnote should have been listed as follows: 1. The […]
LikeLike